
- May. 23, 2025
- Home
- About Us
- Editorial Board
- Instruction
- Subscription
- Advertisement
- Contact Us
- Chinese
- RSS

Chinese Journal of Magnetic Resonance ›› 2021, Vol. 38 ›› Issue (2): 164-172.doi: 10.11938/cjmr20202841
• Articles • Previous Articles Next Articles
Jin-bo YU1,2,Cai ZHANG1,2,Ze-ting ZHANG1,Guo-hua XU1,*( ),Cong-gang LI1
),Cong-gang LI1
Received:2020-07-07
Online:2021-06-05
Published:2020-09-01
Contact:
Guo-hua XU
E-mail:guohua_xu@wipm.ac.cn
CLC Number:
Jin-bo YU,Cai ZHANG,Ze-ting ZHANG,Guo-hua XU,Cong-gang LI. Interactions Between α-synuclein and Intact Mitochondria Studied by NMR[J]. Chinese Journal of Magnetic Resonance, 2021, 38(2): 164-172.

Fig.1
The analysis of isolated mitochondrial purity and viability from the liver of rat. (a) The COX6c component and (b) SOD1 component were detected by western blotting (SOD1 standard sample as positive control in figure (b)), showing the mitochondrial purity. (c)~(e) Mitochondrial viability characterized by fluorescence confocal microscopy: (c) Bright field; (d) Isolated mitochondria were stained with Mito Tracker® Red CMXRos whose accumulation is dependent upon the mitochondrial membrane potential (MMP); (e) Overlay of figure (c) and figure (d)
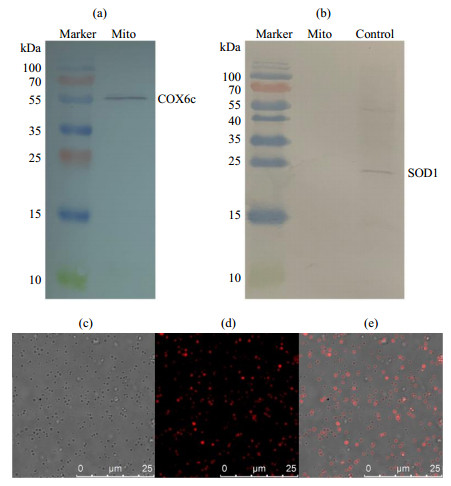

Fig.2
1H-15N SOFAST-HMQC spectra of α-synuclein under different conditions. (a) The amino acid sequence of α-synuclein, the N-terminus region (1~60) are colored in red; (b) Spectral overlay of α-synuclein in absence (blue) and presence (red) of intact mitochondria, the partial peaks of which signal intensity attenuated in the presence of intact mitochondria are marked; (c) Spectral overlay of α-synuclein with (green) and without (blue) PMSF(1 mmol/L) in diluted solution; (d) Spectral overlay of α-synuclein with (cyan) and without (red) PMSF (1 mmol/L) in the presence of intact mitochondria
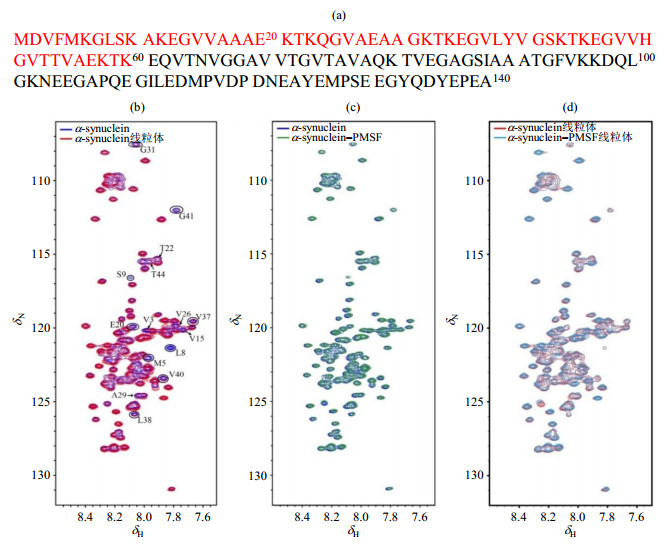
Fig.3
The analysis of residue resolved NMR signal intensity and chemical shift change of α-synuclein in the presence of mitochondria (the values of NMR signal intensity ratio and chemical shift changes are shown as blank for the residues whose signal intensity can not be accurately obtained due to the overlap of spectral peaks). (a) NMR signal intensity ratio (Iα-synuclein-mito/Iα-synuclein) of α-synuclein in the presence and absence of mitochondria; (b) Chemical shift changes of α-synuclein in presence of mitochondria compared to α-synuclein; (c) NMR signal intensity ratio (Iα-synuclein-PMSF-mito/Iα-synuclein) of α-synuclein with PMSF in the presence of mitochondria and α-synuclein; (d) Chemical shift changes of α-synuclein with PMSF in the presence of mitochondria compared to α-synuclein
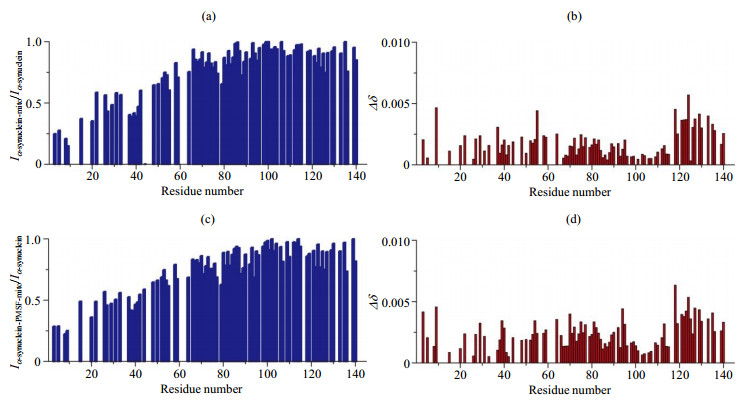
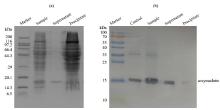
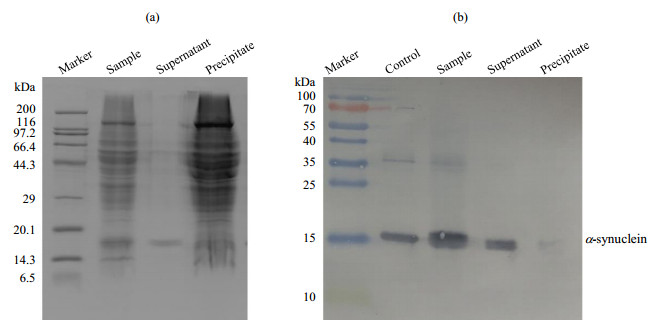
Fig.4
(a) SDS-PAGE and (b) Western-blotting analysis of α-synuclein with PMSF in the presence of intact mitochondria (a) SDS-PAGE analysis showing integrity of mitochondria; (b) Western-blotting analysis showing that α-synuclein is undegraded; Control: α-synuclein standard sample, Sample: α-synuclein with PMSF in the presence of intact mitochondria, Supernatant: the supernatant of the sample (α-synuclein with PMSF in the presence of intact mitochondria) that was centrifuged, Precipitate: the precipitate of the sample that was centrifuged
| 1 |
POLYMEROPOULOS M H , LAVEDAN C , LEROY E , et al. Mutation in the alpha-synuclein gene identified in families with Parkinson's disease[J]. Science, 1997, 276 (5321): 2045- 2047.
doi: 10.1126/science.276.5321.2045 |
| 2 |
SPILLANTINI M G , SCHMIDT M L , LEE V M Y , et al. Alpha-synuclein in Lewy bodies[J]. Nature, 1997, 388 (6645): 839- 840.
doi: 10.1038/42166 |
| 3 |
MASLIAH E , ROCKENSTEIN E , VEINBERGS I , et al. Dopaminergic loss and inclusion body formation in alpha-synuclein mice: Implications for neurodegenerative disorders[J]. Science, 2000, 287 (5456): 1265- 1269.
doi: 10.1126/science.287.5456.1265 |
| 4 |
FINK A L . The aggregation and fibrillation of alpha-synuclein[J]. Accounts Chem Res, 2006, 39 (9): 628- 634.
doi: 10.1021/ar050073t |
| 5 |
ZAMMIT V A , RAMSAY R R , BONOMINI M , et al. Carnitine, mitochondrial function and therapy[J]. Adv Drug Deliver Rev, 2009, 61 (14): 1353- 1362.
doi: 10.1016/j.addr.2009.04.024 |
| 6 |
PALADE G E . The fine structure of mitochondria[J]. Anat Record, 1952, 114 (3): 427- 451.
doi: 10.1002/ar.1091140304 |
| 7 |
FREY T G , MANNELLA C A . The internal structure of mitochondria[J]. Trends Biochem Sci, 2000, 25 (7): 319- 324.
doi: 10.1016/S0968-0004(00)01609-1 |
| 8 |
LIN M T , CANTUTI-CASTELVETRI I , ZHENG K , et al. Somatic mitochondrial DNA mutations in early Parkinson and incidental Lewy body disease[J]. Ann Neurol, 2012, 71 (6): 850- 854.
doi: 10.1002/ana.23568 |
| 9 |
SHAVALI S , BROWN-BORG H M , EBADI M , et al. Mitochondrial localization of alpha-synuclein protein in alpha-synuclein overexpressing cells[J]. Neurosci Lett, 2008, 439 (2): 125- 128.
doi: 10.1016/j.neulet.2008.05.005 |
| 10 |
LI L , NADANACIVA S , BERGER Z , et al. Human A53T alpha-synuclein causes reversible deficits in mitochondrial function and dynamics in primary mouse cortical neurons[J]. PloS ONE, 2013, 8 (12): e85815.
doi: 10.1371/journal.pone.0085815 |
| 11 |
PERFEITO R , LAZAR D F , OUTEIRO T F , et al. Linking alpha-synuclein phosphorylation to reactive oxygen species formation and mitochondrial dysfunction in SH-SY5Y cells[J]. Mol Cell Neurosci, 2014, 62, 51- 59.
doi: 10.1016/j.mcn.2014.08.002 |
| 12 |
GANJAM G K , BOLTE K , MATSCHKE L A , et al. Mitochondrial damage by alpha-synuclein causes cell death in human dopaminergic neurons[J]. Cell Death Dis, 2019, 10 (11): 865- 879.
doi: 10.1038/s41419-019-2091-2 |
| 13 |
SCHAPIRA A H V . Mitochondria in the aetiology and pathogenesis of Parkinson's disease[J]. Lancet Neurol, 2008, 7 (1): 97- 109.
doi: 10.1016/S1474-4422(07)70327-7 |
| 14 |
MIZUNO Y , IKEBE S , HATTORI N , et al. Role of mitochondria in the etiology and pathogenesis of Parkinson's disease[J]. Biochim Biophys Acta, 1995, 1271 (1): 265- 274.
doi: 10.1016/0925-4439(95)00038-6 |
| 15 |
ZIGONEANU I G , YANG Y J , KROIS A S , et al. Interaction of alpha-synuclein with vesicles that mimic mitochondrial membranes[J]. BBA-Biomembranes, 2012, 1818 (3): 512- 519.
doi: 10.1016/j.bbamem.2011.11.024 |
| 16 |
ROBOTTA M , GERDING H R , VOGEL A , et al. Alpha-synuclein binds to the inner membrane of mitochondria in an alpha-helical conformation[J]. Chembiochem, 2014, 15 (17): 2499- 2502.
doi: 10.1002/cbic.201402281 |
| 17 |
ROBOTTA M , HINTZE C , SCHILDKNECHT S , et al. Locally resolved membrane binding affinity of the N-terminus of alpha-synuclein[J]. Biochemistry, 2012, 51 (19): 3960- 3962.
doi: 10.1021/bi300357a |
| 18 | GAO D L , SUN P , WANG Q W , et al. Interactions between albumin and fatty acids studied by NMR spectroscopy[J]. Chinese J Magn Reson, 2018, 35 (3): 338- 344. |
| 高东莉, 孙鹏, 王倩文, 等. 运用NMR研究白蛋白与脂肪酸的相互作用[J]. 波谱学杂志, 2018, 35 (3): 338- 344. | |
| 19 | NING C F , MA M J , GUO C H , et al. Interactions between 5-fluorouracil and PAMAM dendrimers studies by NMR spectroscopy[J]. Chinese J Magn Reson, 2019, 36 (4): 555- 562. |
| 宁彩芳, 马敏珺, 郭朝晖, 等. PAMAM树状大分子与5-氟尿嘧啶相互作用的NMR研究[J]. 波谱学杂志, 2019, 36 (4): 555- 562. | |
| 20 |
THEILLET F X , BINOLFI A , BEKEI B , et al. Structural disorder of monomeric alpha-synuclein persists in mammalian cells[J]. Nature, 2016, 530 (7588): 45- 50.
doi: 10.1038/nature16531 |
| 21 |
ZHENG W W , ZHANG Z T , YE Y S , et al. Phosphorylation dependent alpha-synuclein degradation monitored by in-cell NMR[J]. Chem Commun, 2019, 55 (75): 11215- 11218.
doi: 10.1039/C9CC05662A |
| 22 |
YE Y S , LIU X L , XU G H , et al. Direct observation of Ca2+-induced calmodulin conformational transitions in intact xenopus laevis oocytes by 19F NMR spectroscopy[J]. Angew Chem Int Edit, 2015, 54 (18): 5328- 5330.
doi: 10.1002/anie.201500261 |
| 23 |
XU G H , YE Y S , LIU X L , et al. Strategies for protein NMR in escherichia coli[J]. Biochemistry, 2014, 53 (12): 1971- 1981.
doi: 10.1021/bi500079u |
| 24 |
YE Y S , LIU X L , CHEN Y H , et al. Labeling strategy and signal broadening mechanism of protein NMR spectroscopy in xenopus laevis oocytes[J]. Chemistry, 2015, 21 (24): 8686- 8690.
doi: 10.1002/chem.201500279 |
| 25 |
YE Y S , LIU X L , ZHANG Z T , et al. 19F NMR spectroscopy as a probe of cytoplasmic viscosity and weak protein interactions in living cells[J]. Chem-Eur J, 2013, 19 (38): 12705- 12710.
doi: 10.1002/chem.201301657 |
| 26 |
HOYER W , ANTONY T , CHERNY D , et al. Dependence of alpha-synuclein aggregate morphology on solution conditions[J]. J Mol Biol, 2002, 322 (2): 383- 393.
doi: 10.1016/S0022-2836(02)00775-1 |
| 27 | DAI C Y , LIU M L , LI C G . Salt content-dependent conformational changes of alpha-synuclein studied by 19F NMR[J]. Chinese J Magn Reson, 2015, 32 (1): 33- 44. |
| 戴晨晔, 刘买利, 李从刚. 低盐和高盐环境下α-synuclein构象的19F NMR研究[J]. 波谱学杂志, 2015, 32 (1): 33- 40. | |
| 28 |
COLLOMBET J M , WHEELER V C , VOGEL F , et al. Introduction of plasmid DNA into isolated mitochondria by electroporation[J]. J Biol Chem, 1997, 272 (8): 5342- 5347.
doi: 10.1074/jbc.272.8.5342 |
| 29 |
PARIHAR M S , PARIHAR A , FUJITA M , et al. Mitochondrial association of alpha-synuclein causes oxidative stress[J]. Cell Mol Life Sci, 2008, 65 (7-8): 1272- 1284.
doi: 10.1007/s00018-008-7589-1 |
| 30 |
SCHMITT S , SCHULZ S , SCHROPP E M , et al. Why to compare absolute numbers of mitochondria[J]. Mitochondrion, 2014, 19, 113- 123.
doi: 10.1016/j.mito.2014.06.005 |
| 31 |
MIWA S , TREUMANN A , BELL A , et al. Carboxylesterase converts Amplex red to resorufin: Implications for mitochondrial H2O2 release assays[J]. Free Radical Biol Med, 2016, 90, 173- 183.
doi: 10.1016/j.freeradbiomed.2015.11.011 |
| 32 |
ZHANG Z T , DAI C Y , BAI J , et al. Ca2+ modulating alpha-synuclein membrane transient interactions revealed by solution NMR spectroscopy[J]. BBA-Biomembranes, 2014, 1838 (3): 853- 858.
doi: 10.1016/j.bbamem.2013.11.016 |
| 33 |
FUSCO G , DE SIMONE A , GOPINATH T , et al. Direct observation of the three regions in alpha-synuclein that determine its membrane-bound behaviour[J]. Nat Commun, 2014, 5, 3827- 3834.
doi: 10.1038/ncomms4827 |
| 34 |
ZHANG Z T , JIANG X , XU D R , et al. Calcium accelerates SNARE-mediated lipid mixing through modulating alpha-synuclein membrane interaction[J]. BBA-Biomembranes, 2018, 1860 (9): 1848- 1853.
doi: 10.1016/j.bbamem.2018.03.025 |
| [1] | Chong-wu WANG,Xi HUANG,Lei SHI,Shi-zhen CHEN,Xin ZHOU. Cathepsin B Triggered Hyperpolarization 129Xe MRI Probe for Ultra-Sensitive Lung Cancer Cells Detection [J]. Chinese Journal of Magnetic Resonance, 2021, 38(3): 336-344. |
| [2] | Jia-min WU,Yu-cheng HE,Zheng XU,Yan-he ZHU,Wen-zheng JIANG. A Wide-Band Matching Method for Radio Frequency Coils Used in Soil Moisture Measurement [J]. Chinese Journal of Magnetic Resonance, 2021, 38(3): 414-423. |
| [3] | Zi-hao WANG,He XU,Tao WANG,Shan-zhong YANG,Yun-sheng DING,Hai-bing WEI. NMR Spectroscopic Studies on (exo, endo) C-2 Monosubstituted Norbornene Derivatives [J]. Chinese Journal of Magnetic Resonance, 2021, 38(3): 323-335. |
| [4] | Xin-yi ZHAO,Dong HAN,Hong-jun LUO,Wen-bin SHEN,Gong-jun YANG. Spectroscopic Studies of Delafloxacin Meglumine [J]. Chinese Journal of Magnetic Resonance, 2021, 38(2): 268-276. |
| [5] | Xiao-wen CHEN,Bi-ling HUANG,Shao-hua HUANG,Yu-fen ZHAO. An NMR Study on the clpC Operon Binding Region of Transcription Factor CtsR from Bacillus subtilis [J]. Chinese Journal of Magnetic Resonance, 2021, 38(2): 155-163. |
| [6] | Yi LI,Jia-xiang XIN,Jia-chen WANG,Da-xiu WEI,Ye-feng YAO. Preparation Efficiency of Nuclear Spin Singlet State: A Comparison Among Three Pulse Sequences [J]. Chinese Journal of Magnetic Resonance, 2021, 38(2): 227-238. |
| [7] | Wei ZHANG,Yi-ming WU,Wei-ping CUI,Liang XIAO. Correction for the Nuclear Magnetic Resonance Porosity in Heavy Oil-bearing Reservoirs [J]. Chinese Journal of Magnetic Resonance, 2021, 38(2): 204-214. |
| [8] | Kun MENG,Sheng-jian WANG,Zong-an XUE,Rui-qing HOU,Liang XIAO. Quantitative Evaluation of Shale Pore Structure Using Nuclear Magnetic Resonance Data [J]. Chinese Journal of Magnetic Resonance, 2021, 38(2): 215-226. |
| [9] | Zhi-wu ZHANG,Ju YANG,Ze-feng NIE,Shang-xiang YE,Xu DONG,Chun TANG. Development of a Temperature Senor Based on 19F-labeled Phosphorylated Ubiquitin [J]. Chinese Journal of Magnetic Resonance, 2021, 38(2): 173-181. |
| [10] | LIAO Huai-yu, HAN Hong-yuan, CHEN Fei, ZHANG Hai-yan, YANG Jing, ZHAO Tian-zeng. An NMR Study on Two New β-Dihydroagrofuran Compounds in Celastrus angulatus Maxim [J]. Chinese Journal of Magnetic Resonance, 2021, 38(1): 101-109. |
| [11] | WANG Rui-di, XU Bei-bei, SONG Yan-hong, WANG Xue-lu, YAO Ye-feng. Methanol-Water Interaction in Photocatalytic Methanol Reforming ─ An Operando NMR Study [J]. Chinese Journal of Magnetic Resonance, 2021, 38(1): 43-57. |
| [12] | KE Han-ping, CAI Hong-hao. High-Resolution Localized NMR Spectroscopy Based on Hadamard-Encoding [J]. Chinese Journal of Magnetic Resonance, 2020, 37(4): 524-532. |
| [13] | LIU Si, AN Yan-peng, TANG Hui-ru. Effects of Lyophilization on the Metabonomic Phenotypes of Human Biofluids Characterized with NMR Analysis [J]. Chinese Journal of Magnetic Resonance, 2020, 37(4): 484-489. |
| [14] | LI Ying-jun, YANG Hong-jing, LIU Ji-hong, JIN Kun, LIN Le-di, LIU Xue-jie. Assignments of NMR Spectral Data of a Novel Carbazole-Triazinoindole Based N-Acylhydrazone Derivative [J]. Chinese Journal of Magnetic Resonance, 2020, 37(4): 496-504. |
| [15] | ZHAN Jia-ying, TU Zhang-ren, DU Xiao-feng, YUAN Bin, GUO Di, QU Xiao-bo. Progresses on Low-Rank Reconstruction for Non-Uniformly Sampled NMR Spectra [J]. Chinese Journal of Magnetic Resonance, 2020, 37(3): 255-272. |
| Viewed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
Full text 263
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract 236
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||